Executive Summary
SkinStim Profile
SkinStim is developing what it believes will be the efficacious technology for total and complete skin regeneration based on our proprietary SS-15 skin regeneration composition and patented bioelectric stem cell homing signaling, proliferation and differentiation control and re-starting expression of tropoelastin for elasticity improvement as well bioelectric controlled expression of over a dozen other skin regeneration promoting proteins.
Market
Over one billion people worldwide seek out solutions to help their facial and neck skin appear more youthful. The skin care products market is valued at $177 billion annually. The product for skin burn wound recovery is estimated to be $1.6 billion.

Technology
SkinStim is the first and only skin regeneration technology platform that combines precise regeneration bioelectric signaling and biologics. The company has patented or patent pending over 20 bioelectric signaling sequences to promote skin regeneration including a stem cell homing signal, IGF1 and PDGF expression and controlled expression of tropoelastin. In nearly all humans they lose their ability to express tropoelastin that creates lasting elasticity in our skin in tissues in early puberty. SkinStim has patent pending the only known bioelectric signaling sequence to turn the tropoleastin expression switch back on! Our propriety SS-15 skin regeneration composition is comprised of adipose-derived stem cells and stromal fraction, exosomes, Micro RNA gel, PRF, amniotic fluid, oxygenated nanoparticles, selected alkaloids, selected growth factors including IGF1, nutrient hydrogel and skin matrix. We try to repair all skin defects with non-invasive bioelectric signaling alone and only revert to biologics and the SS-15 composition in the most severe skin regeneration cases.
Advantages
- Stem cell homing to rebuild new skin.
- Tropoelastin release to increase elasticity.
- IGF1 release for DNA repair.
- Combination of bioelectics and biologics for more complete skin regeneration.
Development Stage
- Entering clinical studies now in Los Angeles, Utah, South Africa and Pittsburgh. Expect data on at least 30 to 100 patients by Spring. Plan is to exit to a strategic partner at that time.
Patents
- Over 30 issued patent claims and over 100 new claims pending. Patent and patent pending claims covering stem cell homing control, stem cell differentiation control, tropoelastin release, IGF1 release, PDGF release.
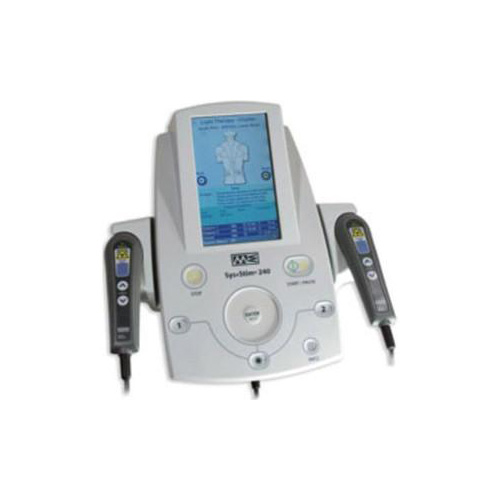
System Components
- In-clinic high precision bioelectric stimulator – FDA 510K market cleared – $3500
- @ home portable stimulator – $300
- PRF bedside processor – $1500
- DermaPen microneedle delivery system – $130
- Ambient temperate amniotic fluid – $600
- SS-15 skin regeneration composition – $3000
- Bioelectric and LED Light Face and Neck Mask – $80
- Super nutrient-infused hydrogel skin gel/cream – $125
Select Publications
See – https://skinstim.lionhearthealthstim.com//related-articles/
Frequently Asked Questions
See – FAQ – Skinstim https://skinstim.lionhearthealthstim.com//faq/
Team
- Howard Leonhardt, Executive Chairman & CEO – 30 years experience. Inventor with over 21 issued U.S. patents and dozens pending. Developed and brought to market leadership and exit multiple healthcare related products.
- Dr. Leslie Miller, Chief Medical Officer – 241 peer reviewed publications, help manage over 80 clinical trials, former Chairman of Cardiovascular Medicine at University of Minnesota for over a decade. Co-Editor of leading stem cell regeneration related textbook.
- Chris Rosgen President – Over 20 years experience in developing healthcare companies especially in aesthetics sector. Producer American Health Journal television program.
- Jeremy Koff, Vice President Business Development – Over 25 yers experience developing life science products.
- Alex Richardson, Vice President of Engineering & Product Development – Over 25 years experience developing and bringing to market life science and precision electronics products. Many years serving Alfred Mann Cos. in Los Angeles.
- Dr. Brett Burton, Director of R&D – Ph.D. Bioengineering U of Utah, 7 years experience in healthcare product development and research.
- Tom Newman, VP of Physician Relations – 30 years healthcare sector sales and marketing experience.
- Dr. Stuart Williams, Chief Advisor Regenerative Therapies – Over 30 years experience organ regeneration research.
- Dr. Jorge Genovese, Vice President Bioelectric Regeneration Research – Over 25 years experience bioelectric and biologics organ regeneration research experience.
- Dr. Richard Miron, Chief Advisor Biologics – 20 years biologics research experience
Facilities
- Dedicated R&D lab space at BioInnovations Gateway
- Dedicated R&D lab space at Center for Medical Innovation
- Shared lab space at USTAR & BioInnovations Gateway
- Shared lab space at Pacific Neurosciences Institute
- Shared lab and office space at LifeX Labs
- WeWork Playa Vista Office Space
- WeWork Brisbane, Australia Office Space
- WeWork Pasadena, CA Office Space
- Shared lab space at University of Northern California Science & Technology Innovation Center (UNC STIC)
- C&S Business Incubator Office Space
- SaltMine CoWorking Office Space
Finance
- 3,000,000 authorized shares.
- Raising $500,000
- $500K takes us through publishing first-in-man results and exit and additional patent filings.
- Target valuation at exit > $300 million down with additional milestone payments and a 3% royalty on net sales.

